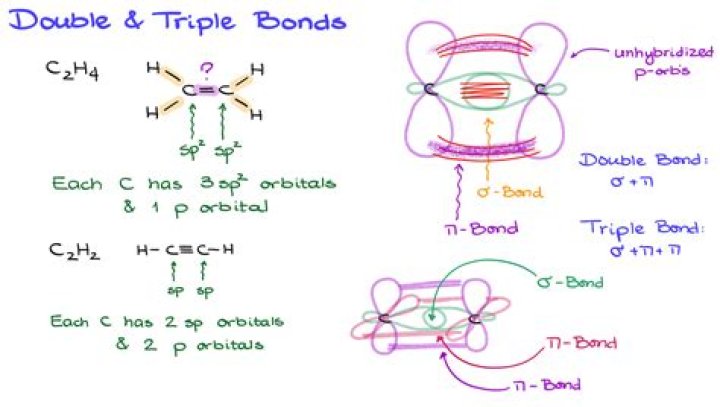
What are lobes in chemistry
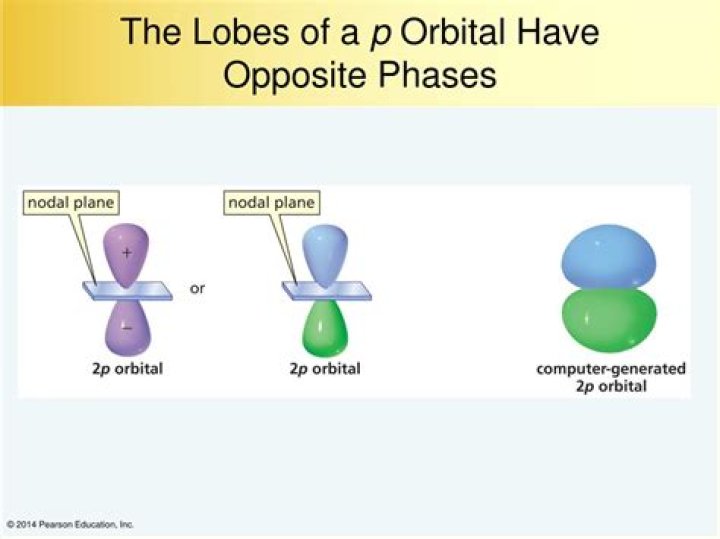
A lobe refers to a high probability density area of finding an electron. A 2p orbital has 2 lobes, most 3d orbitals have 4 lobes with the exception of 3dz^2 which has 3. A radial node (or any node) is a area with zero probability density of finding an electron, for example inside the nucleus.
What are lobes in orbitals?
Visualizing Electron Orbitals The number of possible values is the number of lobes (orbitals) there are in the s, p, d, and f subshells. As shown in Table 1, the s subshell has one lobe, the p subshell has three lobes, the d subshell has five lobes, and the f subshell has seven lobes.
How many lobes are in the d orbital?
Note: Generally, d-orbital has four lobes and 2 nodal planes.
What is 1s 2s 2p 3s 3p?
In the question 1s 2s 2p 3s 3p represents electron orbital energy levels. … The sequence of orbital energy levels is as always-1s < 2s = 2p < 3s = 3p = 3d <4s = 4p = 4d= 4f. The orbital having the same energy is called a degenerate orbital.What is a lobe quantum?
Lobes refers to the shape of electron waves and the area of highest probability of where that electron as a particle would be found.
How many orbitals are in 4f?
For any atom, there are seven 4f orbitals. The f-orbitals are unusual in that there are two sets of orbitals in common use.
What are angular nodes and radial nodes?
Angular nodes are either x, y, and z planes where electrons aren’t present while radial nodes are sections of these axes that are closed off to electrons. where R(r) is the radial component which depends only on the distance from the nucleus and Y(θ,ϕ) is the angular component.
What is the SPDF?
The spdf stands for sharp, principal, diffuse, and fundamental respectively. These letters are used as the visual impression to describe the fine structure of the spectral lines that occurs due to the spin orbital interaction.What is the SPDF notation?
s, p, d, f and so on are the names given to the orbitals that hold the electrons in atoms. These orbitals have different shapes (e.g. electron density distributions in space) and energies (e.g. 1s is lower energy than 2s which is lower energy than 3s; 2s is lower energy than 2p).
Where is 5f on the periodic table?In order to make the overall shape of the table more compact and convenient to display, scientists have adopted the convention of removing the elements with atomic number 57 through 70 and 89 through 102 (the latter being the 5f portion of the table) from their natural position between the s and d blocks and placing …
Article first time published onWhere are the lobes located?
The four lobes of the brain are the frontal, parietal, temporal, and occipital lobes (Figure 2). The frontal lobe is located in the forward part of the brain, extending back to a fissure known as the central sulcus. The frontal lobe is involved in reasoning, motor control, emotion, and language.
Which of the following example orbital have four lobes?
the d-orbitals have (i) four lobes and two nodes (ii) four lobe… A square planar complex is formed by hybridization of which atomic orbitals [ AIEEE 202] a)s, px, p…
How many angular nodes will each F orbital have?
Following this trend, d orbitals would have l = 2 and f orbitals would have l = 3, as they have two and three angular nodes, respectively. Note: As pictured above, the s orbital has uninterrupted electron density all-around, so l = 0.
What are the 7 orbitals?
The shape of the seven 7f orbitals (cubic set). From left to right: (top row) 7fy 3, 7fz 3, 7fx 3, (middle row) 7fy(z 2-x 2), 7fz(x 2-y 2), and 7fx(z 2-y 2) (bottom row) 7fxyz. For each, the green zones are where the wave functions have positive values and the white zones denote negative values.
What is the highest energy orbital?
The order of the electron orbital energy levels, starting from least to greatest, is as follows: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p. Since electrons all have the same charge, they stay as far away as possible because of repulsion.
What is nodal point in chemistry?
Nodal points are only present in p and d orbitals as it is the point at which the shape of the orbital is formed. Nodal planes are the regions around the nucleus where the probability of finding electrons is likely to be zero.
What is radial and angular?
The spherical surfaces around the nucleus where the probability of finding an electron is zero are called radial nodes. The planes or planar areas around the nucleus where the probability of finding an electron is zero are called angular nodes.
What is radial node class 11?
The region where the probability density function reduces to zero is called radial nodes or Nodal surfaces or nodes. For s orbitals, the number of radial nodes increases with the value of the principal quantum number n and found to be equal to (n – 1).
What is the difference between angular and radial nodes?
There are two types of nodes as radial nodes and angular nodes. The main difference between radial nodes and angular nodes is that radial nodes are spherical whereas angular nodes are typically flat planes.
How many electrons are in a 4p orbital?
As each orbital can hold maximum two electrons so total electrons p orbital can hold is 6. Therefore, the 4p orbital can hold two electrons and the 4p subshell can hold a total of six electrons.
Why there is no 3f orbital?
In the second shell, both 2s and 2p orbitals exist, as it can have a maximum of 8 electrons. In the third shell, only the 3s, 3p and 3d orbitals exist, as it can hold a maximum of 18 electrons. Therefore, the 3f orbitals do not exist.
How many nodes are in the 4f orbital?
There are 3 nodes in the 4f orbitals.
What is the highest energy level?
Energy LevelSublevelsMaximum Number of Electrons per Energy Level1s22s8p3s18
How do you write the electron configuration for sodium?
Therefore the sodium electron configuration will be 1s22s22p63s1. The configuration notation provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of an atom.
What are the four quantum numbers?
In atoms, there are a total of four quantum numbers: the principal quantum number (n), the orbital angular momentum quantum number (l), the magnetic quantum number (ml), and the electron spin quantum number (ms).
Why are shells called KLM?
The names of the electron shell were given by a spectroscopist named Charles G Barkla. He named the innermost shell has k shell because he noticed that the X-rays emitted two types energies. … Therefore, he named the innermost shell as the K shell.
What is the p sublevel?
The p sublevel has 3 orbitals, so can contain 6 electrons max. The d sublevel has 5 orbitals, so can contain 10 electrons max. And the 4 sublevel has 7 orbitals, so can contain 14 electrons max. … The p sublevels are called 2p, 3p, and 4p.
What are f block elements called?
The f block elements are the lanthanides and actinides and are called the inner transition elements because of their placement in the periodic table due to their electron configurations.
What is the element name for RA?
radium (Ra), radioactive chemical element, the heaviest of the alkaline-earth metals of Group 2 (IIa) of the periodic table. Radium is a silvery white metal that does not occur free in nature.
How many lobes are in the cerebrum?
Each side of your brain contains four lobes. The frontal lobe is important for cognitive functions and control of voluntary movement or activity. The parietal lobe processes information about temperature, taste, touch and movement, while the occipital lobe is primarily responsible for vision.
What are the 4 lobes of the cerebrum and their functions?
The four lobes include the occipital, temporal, frontal, and parietal lobes. Each lobe is responsible for a specific task. The frontal lobe functions in solving problems, controlling body movements, sentence formation, and personality traits. The occipital lobe functions in processing visual images.