What is a reverse dot blot
The reverse dot-blot method is a simple and rapid diagnostic procedure that allows screening of sample for a variety of mutations/polymorphisms in a single hybridization reaction. Several methods of immobilizing the oligonucleotide probes are discussed.
What is the difference between dot blot and western blot?
A dot blot (or slot blot) is a technique in molecular biology used to detect proteins. It represents a simplification of the western blot method, with the exception that the proteins to be detected are not first separated by electrophoresis. … However, it offers no information on the size of the target protein.
What is reverse dot blot hybridization?
Reverse dot blot hybridization assays involve the amplification and labeling of the DNA sequence of interest (the target DNA), followed by hybridization of the labeled amplicon to oligonucleotides (the probes) immobilized on a membrane.
What is the dot blot technique?
Dot blot is a technique for detecting, analyzing, and identifying proteins, similar to the western blot technique, but differing in that protein samples are not separated electrophoretically but are spotted through circular templates directly onto the membrane or paper substrate.What is slot blot hybridization?
The dot-blot hybridization is a nucleic acid hybridization technique where complementary single-stranded sequences of the probe (either RNA or DNA) hybridizes with single-stranded sequences of the test samples (either RNA or DNA) under suitable conditions of temperature and salt concentration.
Is dot blot more sensitive than Western blot?
The dot-blot assays were therefore apparently more sensitive than Western blot assays; in previous studies using other anti-DENV NS1 glycoprotein-specific MAbs, the maximum sensitivity was obtained with a 10 ng band [7, 21].
What is colony blot?
a procedure in which an imprint of the colonies grown on the surface of a solid nutrient medium is transferred to a membrane and the Cells lysed or disrupted, allowing the DNA or protein to bind to the membrane.
What is dot blot Elisa?
An enzyme-linked immunosorbent assay using nitrocellulose blotting membrane (dot blot ELISA) was developed for the detection of antibodies against Newcastle disease virus (NDV) in chickens. In this method, a nitrocellulose blotting membrane was used as the solid phase carrier.What is a Southern blot test?
Southern blotting is a laboratory technique used to detect a specific DNA sequence in a blood or tissue sample. … The DNA fragments are transferred out of the gel to the surface of a membrane. The membrane is exposed to a DNA probe labeled with a radioactive or chemical tag.
How do you prepare a dot blot sample?- Cut the nitrocellulose membrane into a 2×5 cm rectangle. …
- Serially dilute samples using PBS (pH 7.4).
- Blot 2 µl of each sample into the centre of each grid.
- Allow to dry for 30 min.
- Incubate the membrane in blocking buffer (5% (w/v) non-fat milk in TBST) for 1 hour at room temperature.
What is the difference between nitrocellulose and PVDF membranes?
In general, nitrocellulose membranes offer the lowest membrane autofluorescence, but low-fluorescence PVDF offers higher binding capacity and better tensile strength.
What does Northern blot tell you?
A northern blot is a laboratory method used to detect specific RNA molecules among a mixture of RNA. Northern blotting can be used to analyze a sample of RNA from a particular tissue or cell type in order to measure the RNA expression of particular genes.
Does Western blot use hybridization?
Western blot analysis is based on a protein/protein hybridization technique that is used for immunodetection of specific antigen(s) of interest in a complex mixture of proteins. This is a simple, sensitive, and effective technology that has been used in immunology, molecular and cellular biology, and protein chemistry.
How do you quantify a dot blot?
There are two built in methods for analyzing a dot blot in ImageJ. The first is to treat each row as a horizontal “lane” and use ImageJ’s gel analysis function. The second is to subtract the background and measure the integrated density of each dot.
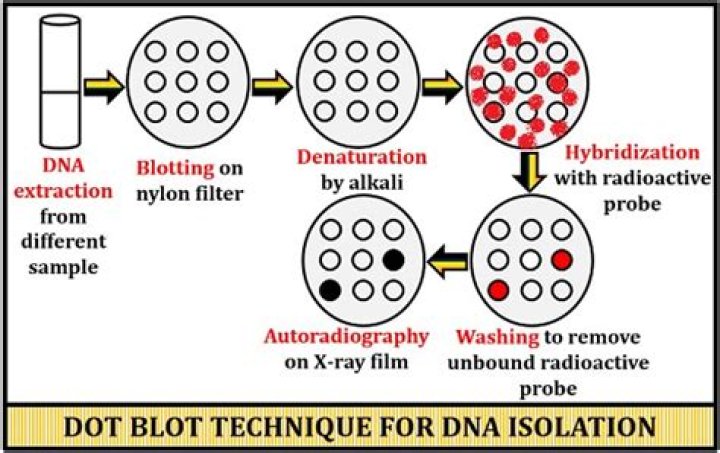
How do you denature DNA for dot blot?
Dot Blot Analysis Denature the isolated DNA (1 mg per sample) in 0.1 M NaOH for 10 min at 95 °C. Neutralize the DNA with 1 M NH4OAc on ice, and then dilute two-fold. Spot 2 µL of the serial diluted genomic DNA on an N+ membrane. Blot the membrane at 80 °C for 30 min.
How do I prevent background signal on my dot Elisa?
- Washing is crucial. …
- Blocking is also (perhaps more) crucial. …
- Optimize antibody concentrations. …
- Be diligent with detection reagents. …
- Survey the whole system for optimal results.
What is the difference between Western Northern and Southern blot?
The western blot is used to detect specific proteins in a sample by “immunoblotting”. … The major difference between the two is that northern blotting is used for RNA detection whereas southern blotting is used for the detection of a specific DNA sequence in large, complex samples of DNA.
Is there an eastern blot?
The eastern blot, or eastern blotting, is a biochemical technique used to analyze protein post-translational modifications including the addition of lipids, phosphates, and glycoconjugates. … In principle, eastern blotting is similar to lectin blotting (i.e., detection of carbohydrate epitopes on proteins or lipids).
What does a western blot tell you?
A western blot is a laboratory method used to detect specific protein molecules from among a mixture of proteins. … Western blots can also be used to evaluate the size of a protein of interest, and to measure the amount of protein expression.
What is the difference between western blot and Elisa?
The key difference between Elisa and western blot is that Elisa or enzyme-linked immunoassay is a diagnostic tool that detects whether the patient has been exposed to a particular type of virus or another infectious agent while western blot is a technique which detects a specific protein from a protein sample.
What is the nitrocellulose membrane?
Nitrocellulose membranes are a popular matrix used in protein blotting because of their high protein-binding affinity, compatibility with a variety of detection methods (chemiluminescence, chromogenic, and fluorescence), and the ability to immobilize proteins, glycoproteins, or nucleic acids.
Which is better nitrocellulose or PVDF?
While nitrocellulose is brittle and fragile, PVDF is more durable and has higher chemical resistance making it ideal for reprobing and sequencing applications. Nitrocellulose can prove to be difficult to strip and reprobe without losing signal.
What are the advantages and disadvantages of PVDF and nitrocellulose membranes?
The good: – Membranes offer higher mechanical strength and allow for re-probing and storage. – Membranes have a higher binding capacity (150-160 µg/cm2) than nitrocellulose (80-100 µg/cm2) [7]. – PVDF is hydrophobic and so lends itself well to the analysis of hydrophobic proteins.
Is nitrocellulose hydrophilic or hydrophobic?
On the other hand, hydrophilic membranes are needed in the application of fast diagnostic products. The post-treatment of the membrane is required because of the hydrophobicity of nitrocellulose.
What is the difference between northern blot and RT PCR?
Although northern blot analysis is effective for quantifying gene expression, reverse transcription-polymerase chain reaction (RT-PCR) is much more sensitive. Obtaining quantitative RT-PCR results, however, can be challenging.
Do people still use Northern blots?
2.6. Northern blotting was used to identify the very first miRNAs and still remains a gold standard for miRNA expression analysis. After isolation of total RNA from cells or tissue, the small RNAs are fractionated by electrophoresis on a high percentage gel.
Who invented Southern blotting?
The technique was named after its inventor, Edward Southern. As a lab procedure, Southern blots can be used to analyze an organism’s total DNA, also known as its genome, in order to identify a specific sequence of interest.
What are the different types of blotting?
- Southern blotting.
- Western blotting.
- Northern blotting.
- Eastern blotting.
Which is the most common ligand in western blotting?
Which is the most common ligand in Western blotting? Explanation: Ligands are used to facilitate protein-ligand interactions in the blotting technique. The most common ligands used are antibodies.
Who invented Western blotting?
The term “western blot” was given by W. Neal Burnette in 1981, although the method itself originated in 1979 in the laboratory of Harry Towbin at the Friedrich Miescher Institute in Basel, Switzerland.