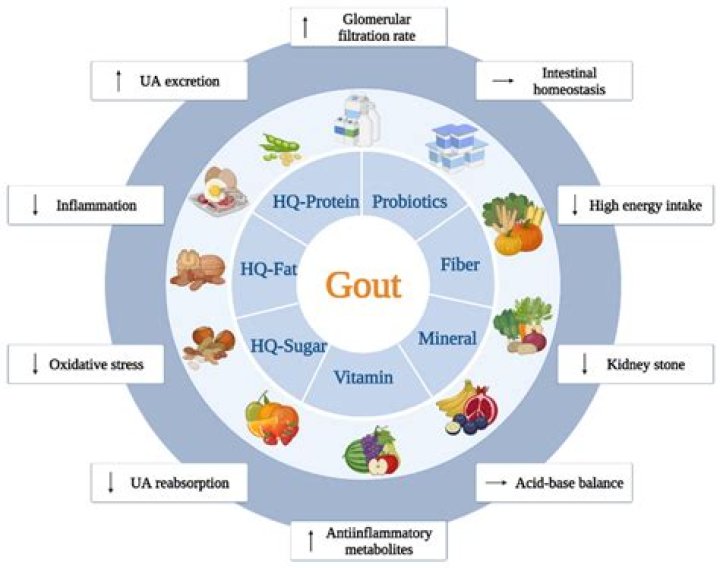
What is a visual indicator
A visual indicator is a “marker” that helps users quickly locate an item that has an important distinctive attribute within a list of otherwise similar objects. Indicators on ecommerce websites are often used to denote exclusive products or items on sale, as seen on the mobile screenshot above of REI.com.
What is a visual indicator what is its relationship to acids and bases?
are the predominate species. The indicator changes color when the pH is between pKa – 1 and pKa + 1, changing from yellow to orange to red (or from red to orange to yellow).
Which of the following is an example of visual indicator?
Alcoholic solution of phenolphthalein (1%) has been frequently applied in titration as a pH visual indicator to reveal information about other compounds, including mineral acids, organic acids, and most alkalis.
What are 3 examples of indicators?
Some examples of indicators are Litmus, turmeric, phenolphthalein, etc. Any material that offers a visual evidence of the presence or absence of a threshold concentration of a chemical species, such as an acid or an alkali in a solution, generally by a colour change are called the chemical indicators.What are visual indicators in Titrimetry?
Visual indicators are widely used for end point detection in the titrimetric analyses. The indicators used normally correspond to the titration reaction and have acid-base or complex- , precipitate formation or oxidation-reduction properties, respectively.
What are the types of indicators in chemistry?
Litmus, phenolphthalein, and methyl orange are all indicators that are commonly used in the laboratory.
What is Visual titration?
A visual and green titrimetric method is proposed for the determination of the acid number of oils and fats. The sample is dissolved in a water–ethanol mixture (1:1 v/v) and titrated with a 0.02 mol L−1 aqueous NaOH solution. Phenolphthalein is used as acid-basic indicator.
What is an indicator write the name of an indicator?
NameAcid ColorpH Range of Color ChangeLitmusRed5.0 – 8.0Bromothymol blueYellow6.0 – 7.6Thymol blueYellow8.0 – 9.6PhenolphthaleinColorless8.2 – 10.0What is an indicator Name three indicators?
Three common indicators are litmus, phenolphthalein and methyl orange.
What are the 4 types of indicators?- Trend indicators. These technical indicators measure the direction and strength of a trend by comparing prices to an established baseline. …
- Momentum indicators. …
- Volatility Indicators. …
- Volume Indicators.
What are 5 types of indicators?
- Input indicators. These indicators refer to the resources needed for the implementation of an activity or intervention. …
- Process and output indicators. Process indicators refer to indicators to measure whether planned activities took place. …
- Outcome indicators. …
- Impact indicators.
What makes a good indicator chemistry?
In all cases, though, a good indicator must have the following properties: The color change must be easily detected. The color change must be rapid. The indicator molecule must not react with the substance being titrated.
What are the examples of artificial indicators?
When we create an indicator in the laboratory using chemicals then it is known as a synthetic or artificial indicator. Some examples of synthetic indicators are phenolphthalein, methyl orange, methyl red, malachite green, etc.
Is a Metallochromic indicator?
Pyrocatecholphthalein has been used as a metallochromic indicator for the direct complexometric titrations of a number of metal ions with EDTA, both in acidic and in alkaline medium.
Which indicator is used in hardness determination?
Eriochrome Black T is a complexometric indicator that is used in complexometric titrations, e.g. in the water hardness determination process. It is an azo dye.
What is a phenolphthalein indicator?
phenolphthalein, (C20H14O4), an organic compound of the phthalein family that is widely employed as an acid-base indicator. As an indicator of a solution’s pH, phenolphthalein is colourless below pH 8.5 and attains a pink to deep red hue above pH 9.0.
What is titration chemistry?
A titration is defined as ‘the process of determining the quantity of a substance A by adding measured increments of substance B, the titrant, with which it reacts until exact chemical equivalence is achieved (the equivalence point)’.
What is titrant chemistry?
Definition of titrant : a substance (such as a reagent solution of precisely known concentration) that is added in titration.
What is the function of an indicator in a titration?
Indicator is the substance that changes or imparts certain color at the stoichiometric end point of the titration. This means when a certain concentration of the acid or a base is completely neutralized, then at the end point we observe a color change. The neutralization between NaOH and HCl is the basis of titration.
Why do we use titration?
The purpose of titration is to determine an unknown concentration in a sample using an analytical method. … When the titration reaches an endpoint, the amount of titrant is recorded and used to calculate the unknown concentration.
What is end point in chemistry?
end point: the point during a titration when an indicator shows that the amount of reactant necessary for a complete reaction has been added to a solution.
What are two types of indicators?
Types of Indicators Artificial and Natural indicators are the two types of Chemical indicators.
What are different types of indicators with example?
An indicator prepared from natural substances is known as natural indicator. Examples are Litmus, Turmeric, China rose petals, snowball plant, Red Cabbage and Grape Juice… . An indicator prepared from artificial substances is known as synthetic indicator Examples are Phenolphthalein and Methyl Orange.
What is an indicator explain types of indicator?
An indicator is a substance that changes its colour in acidic and basic medium.An indicator shows one colour in acidic medium and different colour in basic medium so that they are called indicators. eg:-Litmus , turmeric.
What is an indicator Name 4 natural indicators?
Natural Indicator is a type of indicator that can be found naturally and can determine whether the substance is an acidic substance or a basic substance. Some examples of natural indicators are red cabbage, turmeric, grape juice, turnip skin, curry powder, cherries, beetroots, onion, tomato, etc.
Which is the most common indicator?
Detailed Solution Litmus is the most commonly used indicator in laboratories. Litmus indicator solution turns red in acidic solutions and blue in alkaline solutions.
What is name of the most common indicator used in laboratory?
The most commonly used indicator in the laboratory is universal indicator. A universal indicator is a pH indicator composed of a solution of several compounds that exhibits several smooth colour changes over a pH value range from 1-14 to indicate the acidity or alkalinity of solutions.
How many indicators are there?
There are four main types of technical indicators: Trend Following, Oscillators, Volatility and Support/Resistance.
What is the most accurate indicator?
- Moving Average Convergence Divergence (MACD) …
- Relative Strength Index (RSI) …
- Bollinger Bands. …
- Stochastic Oscillator. …
- On-Balance Volume. …
- Ichimoku Cloud. …
- Fibonacci Retracement Levels. …
- 52-Week High.
Which is the best indicator?
- Stochastic oscillator.
- Moving average convergence divergence (MACD)
- Bollinger bands.
- Relative strength index (RSI)
- Fibonacci retracement.
- Ichimoku cloud.
- Standard deviation.
- Average directional index.
What is an indicator matrix?
The Logical Framework Matrix or Indicator Matrix is a planning tool which in a summarized, simple and harmonic way, clearly establishes a program’s objectives, incorporates the indicators that measure said objectives and its expected results; identifies the means to obtain and verify the indicator information and …