What is cyclic Halonium ion
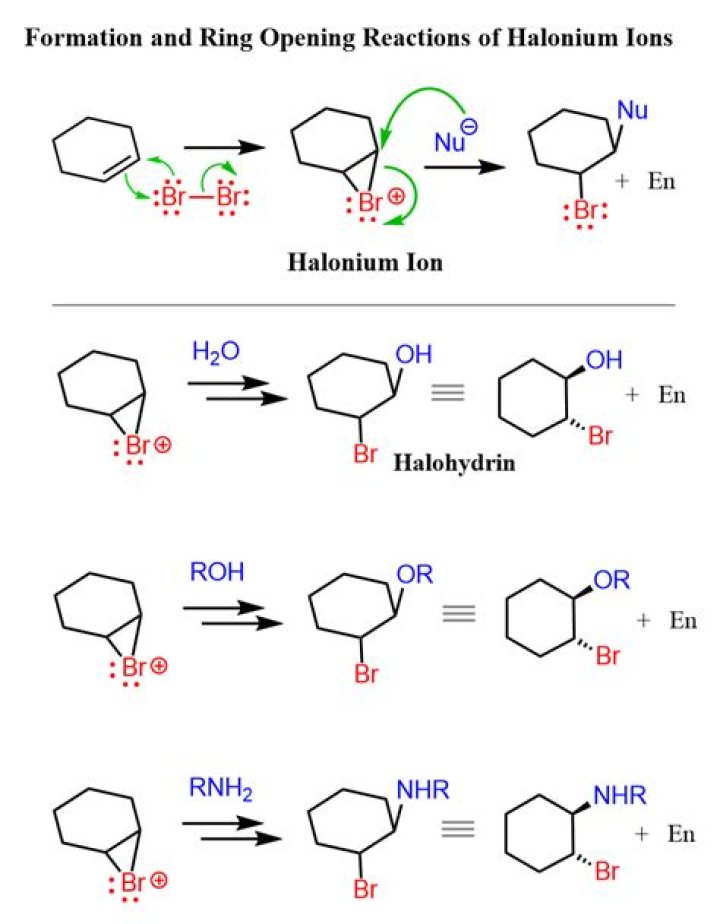
A halonium ion is any onium ion containing a halogen atom carrying a positive charge. … The 3-membered cyclic variety commonly proposed as intermediates in electrophilic halogenation may be called haliranium ions, using the Hantzsch-Widman nomenclature system.
How cyclic Bromonium ion is formed?
The π electrons act as a nucleophile, attacking the bromine, displacing a bromide ion but forming a cationic cyclic bromonium ion as an intermediate.
What is Bromonium ion?
Bromonium ions are cyclic intermediates in electrophilic brominations of C = C double bonds resulting in stereospecific anti additions. Bromonium ions are planar bridging the original π bond of the alkene or alkyne.
What is a halonium ion intermediate?
A halonium ion is a species in which there is a halogen atom that posses an octet of electrons but bears a formal charge of +1. Halonium ions are intermediates in the reaction of alkenes with halogens.Is Bromonium ion a Carbocation?
The bromonium ion is then attacked from the back by a bromide ion formed in a nearby reaction. … In the process, the electrons in the bromine-bromine bond are repelled until they are entirely on the right-hand bromine atom, producing a bromide ion. Exactly as with ethene, a carbocation is formed.
Are bromonium ions stable?
Model #1 – A Stable Bromonium Ion. A bromonium can be stabilized by placing large groups around the alkene. These groups offer steric hindrance to the bromide anion so that backside attack can’t occur.
Why Bromonium ion is more stable than carbocation?
The bromonium (or chloronium) ion formation stabilizes the positive charge and prevents carbocation rearrangement. … Bonding of an electrophilic species to the double bond of an alkene forms preferentially to produce the more stable (more highly substituted) carbocation.
Is a halohydrin an alcohol?
In organic chemistry a halohydrin (also a haloalcohol or β-halo alcohol) is a functional group in which a halogen and a hydroxyl are bonded to adjacent carbon atoms, which otherwise bear only hydrogen or hydrocarbyl groups (e.g. 2-chloroethanol, 3-chloropropane-1,2-diol).What is syn addition and anti addition?
Syn addition is the addition of two substituents to the same side of the unsaturated molecule. Anti addition, on the other hand, is the addition of two substituents in opposite directions.
What is bromination of an alkene?Reaction Overview: The alkene halogenation reaction, specifically bromination or chlorination, is one in which a dihalide such as Cl2 or Br2 is added to a molecule after breaking the carbon to carbon double bond. The halides add to neighboring carbons from opposite faces of the molecule.
Article first time published onIs a Bromonium ion an electrophile?
Furthermore, the Br is acting both as electrophile (accepting a pair of electrons from the π bond) and nucleophile (donating a pair of electrons to the π bond).
Is Bromonium an electrophile?
Bromine molecule undergo heterolytic cleavage to form Br+ and Br− ions. … Whereas the Br+ ion is very unstable and to attain stability it takes part in chemical reaction. Since Br+ wants to gain electron to attain stability, so it is an. Electrophile (an electron loving specie).
What is used for the production of Bromonium Electrophile?
1,B, electrophilic reagents (e.g., the bromonium ion [Br+]), generated by the reaction of diatomic bromine (Br2) and aluminum bromide (AlBr3)], were shown to react with benzene to yield a resonance-stabilized cation called an arenium ion.
What is an electrophile vs Nucleophile?
Electrophiles are those reactants that are either positively charged or neutral with no lone pair of electrons. … A nucleophile is that chemical species that has negative charge or that has lone pairs of electrons. Lone pair of electrons is those electrons that do not get used in the bond.
What is the purpose of electrophilic addition?
Electrophilic addition reactions are an important class of reactions that allow the interconversion of C=C and C≡C into a range of important functional groups including alkyl halides and alcohols. Conceptually, addition is the reverse of elimination (see Chapter 5) which can be used to prepare alkenes.
Why is bromine electrophilic addition?
The electron rich part of the double bond causes a dipole to form meaning that bromine becomes electron deficient and therefore becomes the electrophile (hence electrophillic addition). The double bond attacks the bromine and forms a new carbon – bromine bond, while the other carbon atom becomes electron deficient.
What does NBS mean in organic chemistry?
N-Bromosuccinimide or NBS is a chemical reagent used in radical substitution, electrophilic addition, and electrophilic substitution reactions in organic chemistry.
Is BR a bromine?
Bromine is a chemical element with symbol Br and atomic number 35. Classified as a halogen, Bromine is a liquid at room temperature.
What happens when propene reacts with bromine?
In common with all other alkenes, propene reacts in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. … The bromine loses its original red-brown colour to give a colourless liquid. In the case of the reaction with propene, 1,2-dibromopropane is formed.
How is oxonium ion formed?
Other hydrocarbon oxonium ions are formed by protonation or alkylation of alcohols or ethers (R−C−−R1R2). Secondary oxonium ions have the formula R2OH+, an example being protonated ethers. … It is also used for preparation of enol ethers and related functional groups.
Which of the following carbonium ion is most stable?
The most stable carbonium ion (carbocation) is tertiary carbonium ion.
Is Halohydrin formed by markovnikov?
Reaction Overview: The Halohydrin formation reaction involves breaking a pi bond and creating a halohydrin in its place. … This reaction takes place in water and yields an anti-addition reaction which follows Markovnikov’s rule.
What is syn addition?
Syn addition is the addition of two substituents to the same side (or face) of a double bond or triple bond, resulting in a decrease in bond order but an increase in number of substituents. Generally the substrate will be an alkene or alkyne. … The classical example of this is bromination (any halogenation) of alkenes.
What is Syn anti?
When two atoms are added to the same side, it’s a syn relationship; when two atoms are added to opposite sides it’s an anti relationship.
What is SYN chemistry?
Illustrated Glossary of Organic Chemistry – Syn addition. Syn addition: An addition reaction in which all new bonds are formed on the same face of the reactant molecule.
What are Halohydrins used for?
1. -), also known as haloalcohol dehalogenases, halohydrin epoxidases or halogen-halide lyases, are versatile biocatalysts. These enzymes can be used for the synthesis of important building blocks and precursors in the pharmaceutical, agrochemical and chemical industries.
Why is Halohydrin anti addition?
The stereochemistry of this reaction is anti-addition because the solvent approaches the bromonium ion with back side orientation to produce the addition product.
Does Lindlar's catalyst reduce alkenes?
Lindlar’s catalyst is a palladium catalyst poisoned with traces of lead and quinoline, that reduce its activity such that it can only reduce alkynes, not alkenes.
What bromination means?
Bromination: Any reaction or process in which bromine (and no other elements) are introduced into a molecule. Bromination of an alkene by electrophilic addition of Br2. Bromination of a benzene ring by electrophilic aromatic substitution.
Is bromination syn or anti?
Bromination of Alkenes Gives anti Products Although we saw that several key reactions of alkenes were consistent with this mechanism, it isn’t the case for all.
What is hydration of alkene?
Hydration of Alkenes The net addition of water to alkenes is known as hydration. The result involves breaking the pi bond in the alkene and an OH bond in water and the formation of a C-H bond and a C-OH bond.