What is each atom left with
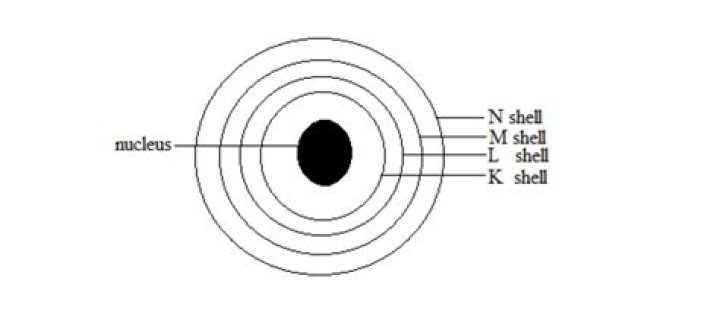
Each atom is left with a complete outer shell. An ionic bond forms between a metal ion with a positive charge and a nonmetal ion with a negative charge.
When an atom loses one of its electrons the atom is left with a?
Forming an Ion An atom that loses one or more valence electrons to become a positively charged ion is known as a cation, while an atom that gains electrons and becomes negatively charged is known as an anion.
What are the 4 types of bonds in chemistry?
There are four types of chemical bonds essential for life to exist: Ionic Bonds, Covalent Bonds, Hydrogen Bonds, and van der Waals interactions.
What is an atomic bond made of?
A strong chemical bond is formed from the transfer or sharing of electrons between atomic centers and relies on the electrostatic attraction between the protons in nuclei and the electrons in the orbitals. The types of strong bond differ due to the difference in electronegativity of the constituent elements.What are ionic bonds formed between?
ionic bond, also called electrovalent bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom.
What happens to an atom when it loses electrons?
Unlike protons, electrons can move from atom to atom. If an atom has an equal number of protons and electrons, its net charge is 0. If it gains an extra electron, it becomes negatively charged and is known as an anion. If it loses an electron, it becomes positively charged and is known as a cation.
What is an atom with a full outer shell called?
A full outer shell is known as the noble gas configuration where the outer shell of an atom is energetically stable and contains 8 outer electrons.
What is holding the atoms together?
Chemical bonds are forces that hold atoms together to make compounds or molecules. Chemical bonds include covalent, polar covalent, and ionic bonds. Atoms with relatively similar electronegativities share electrons between them and are connected by covalent bonds.Do atoms touch?
If “touching” is taken to mean that two atoms influence each other significantly, then atoms do indeed touch, but only when they get close enough. … With 95% of the atom’s electron probability density contained in this mathematical surface, we could say that atoms do not touch until their 95% regions begin to overlap.
Why do atoms combine?Atoms mainly combine to fill the outermost shell and they can combine with similar elements or different elements. Sometimes, they can achieve stability even after half-filled subshell.
Article first time published onWhat is the weakest bond?
The ionic bond is generally the weakest of the true chemical bonds that bind atoms to atoms.
What bond is the strongest?
Covalent bond is the strongest bond. Answer: There are a variety of ways atoms bond to one another. Some bonds are weaker, and some are stronger.
What is the weakest bond in biology?
A Chemical bond is technically a bond between two atoms that results in the formation of a molecule , unit formula or polyatomic ion. The weakest of the intramolecular bonds or chemical bonds is the ionic bond.
What is difference between ionic bond and covalent bond?
Ionic bondsCovalent bondsA polar bond is formed by the attraction between oppositely-charged ions.Usually, an electron is more attracted to one atom than to another, forming a polar covalent bond.
Is NaCl ionic or covalent?
Ionic bonds usually occur between metal and nonmetal ions. For example, sodium (Na), a metal, and chloride (Cl), a nonmetal, form an ionic bond to make NaCl. In a covalent bond, the atoms bond by sharing electrons.
What bonds do nonmetals form?
Nonmetals can form different types of bonds depending on their partner atoms. Ionic bonds form when a nonmetal and a metal exchange electrons, while covalent bonds form when electrons are shared between two nonmetals.
What is Group 18 called?
noble gas, any of the seven chemical elements that make up Group 18 (VIIIa) of the periodic table. The elements are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), radon (Rn), and oganesson (Og).
What is the innermost shell of an atom called?
First Electron Shell The closest orbital to the nucleus, called the 1s orbital, can hold up to two electrons. This orbital is equivalent to the innermost electron shell of the Bohr model of the atom. It is called the 1s orbital because it is spherical around the nucleus.
Does lithium have a full outer shell?
isotope, which has only 3 neutrons. The third electron in lithium cannot fit in the lowest energy shell, which, as noted above, is full with only 2 electrons. Therefore, the third electron in lithium goes into a second shell, that is, an outer shell.
Can an atom lose a proton?
Alpha decay is the only way in which an atom will spontaneously lose protons. An alpha particle consists of two protons and two neutrons. It is essentially the nucleus of a helium atom. After an atom undergoes an alpha emission, it has two fewer protons and becomes an atom of a different element.
Does oxygen gain or lose electrons?
Oxygen is therefore also a really good oxidizing agent, and makes other elements lose electrons, while it gets reduced (gain electrons) itself. Reduction on the other hand, is opposite of oxidation, and is when the atom gains electrons, or loses an oxygen atom.
Is K an anion or cation?
FamilyElementIon NamePotassiumPotassium cationIIABerylliumBeryllium cationMagnesiumMagnesium cationCalciumCalcium cation
Do atoms have Colour?
atoms (as opposed to molecules) do not have colors – they are clear except under special conditions..
Are atoms 99.99 empty space?
Atoms are not mostly empty space because there is no such thing as purely empty space. Rather, space is filled with a wide variety of particles and fields. … It’s true that a large percentage of the atom’s mass is concentrated in its tiny nucleus, but that does not imply that the rest of the atom is empty.
What are the 4 types of atoms?
- Description. Atoms are made of tiny particles called protons, neutrons and electrons. …
- Stable. Most atoms are stable. …
- Isotopes. Every atom is a chemical element, like hydrogen, iron or chlorine. …
- Radioactive. Some atoms have too many neutrons in the nucleus, which makes them unstable. …
- Ions. …
- Antimatter.
What holds everything together?
Gravity is the force that all objects with mass exert upon one another, pulling the objects closer together. … The tiny particles that make up matter, such as atoms and subatomic particles, also exert forces on one another.
Can mixtures be broken down physically?
Note that a mixture: consists of two or more different elements and/or compounds physically intermingled, can be separated into its components by physical means, and. often retains many of the properties of its components.
What three forces hold an atom together?
The “fasteners” are called forces, and there are three different kinds of them at work inside the atom: electromagnetic force, strong nuclear force, and weak nuclear force.
Why do atoms combine 11th?
An atom is the smallest particle of an element. … It is so because the atoms in its outermost valence shell have a maximum of eight electrons and in order to complete this octet, they have a strong tendency to combine among themselves to acquire the stable electronic configuration of the nearest noble gas.
Can atoms exist independently?
Atoms can not exist independently. This is because atoms accept electrons or donate electrons to form stable octet fulfilled electronic configurations and this is only possible by formation of molecules. … But only inert gases already have octet fulfilled configurations so they can exist as atoms.
What happens when atoms are combined together?
When two or more atoms chemically bond together, they form a molecule. … In a covalent bond, electrons are shared between atoms. The bonds between the two hydrogen atoms and the oxygen atom in a molecule of water are covalent bonds. As its name implies, a metallic bond occurs between metallic substances.