What is the Colour of NiCl2
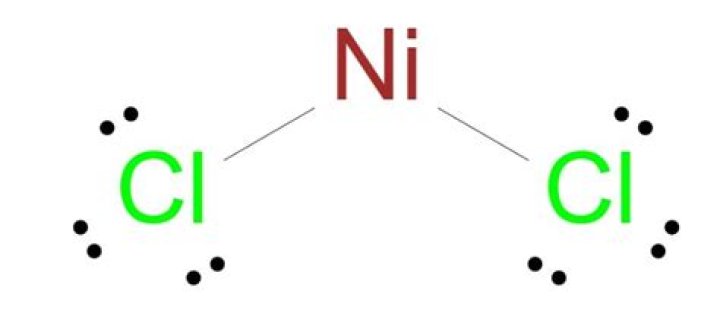
Nickel chloride appears as a brown or green colored solid.
Why is NiCl2 green?
In general, NiCl2 is the most important source of nickel for chemical synthesis, and it is used for electroplating nickel onto other metals 10 (fig. 2). The anhydrous salt of NiCl2 is yellow, but after the addition of H2O to produce the hydrate [Ni(H2O)6]2+, the colour changes to green.
What color is NiCl2?
II) sulfate pentahydrateCuSO4 · 5H2OBlueCobalt(II) chloride hexahydrateCoCl2 · 6H2ODeep magentaManganese(II) chloride tetrahydrateMnCl2 · 4H2OPinkCopper(II) chloride dihydrateCuCl2 · 2H2OBlue-greenNickel(II) chloride hexahydrateNiCl2 · 6H2OGreen
Is nickel chloride a solid?
Nickel chloride is a yellow deliquescent solid with a boiling point of 973°C(1690°F). Nickel chloride is soluble in water and alcohol. Nickel chloride(hydrated),NiCI2·H20, is a gray deliquescent solid that is also soluble in water and alcohol.It is used in nickel plating.Is NiCl2 aqueous or solid?
Nickel chloride is water soluble (642 g l−1 for anhydrous; 2540 g l−1 for hexahydrate) and would be expected to release divalent nickel into the water.
Why is NiCl2 ionic?
NiCl2 adopts the CdCl2 structure. In this motif, each Ni2+ center is coordinated to six Cl− centers, and each chloride is bonded to three Ni(II) centers. In NiCl2 the Ni-Cl bonds have “ionic character”.
What ions are present in NiCl2?
When nickel(II) chloride dissolves in water, it dissociates into nickel ions and chloride ions. The formula for nickel ions is Ni+2 …
What type of electrolyte is NiCl2?
Electrochemical behavior of NiCl2/Ni in acidic AlCl3-based ionic liquid electrolyte.Is NiCl2 polar?
NiCl2 is polar. Electronegativity is the strength with which an atom pulls on shared electrons.
What is a hexahydrate?Definition of hexahydrate : a chemical compound with six molecules of water.
Article first time published onIs cobalt chloride ionic?
NamesChemSpider22708ECHA InfoCard100.028.718EC Number231-589-4PubChem CID3032536
What color is chloride?
The ion is colorless and diamagnetic. In aqueous solution, it is highly soluble in most cases; however, for some chloride salts, such as silver chloride, lead(II) chloride, and mercury(I) chloride, they are only slightly soluble in water.
What is the lambda max for NiCl2?
Med.λmin (nm)λmax (nm)Ar582583
Is NiCl2 a precipitate?
If solid NiCl2 is added to aqueous NaOH, can I see the green precipitate? Yes. … Because solid NiCl2 dissociates to nickel +2 ions and chloride ions in water. Then, nickel +2 ion and hydroxyl ions form nickel hydroxide precipitate.
Is NiCl2 ionic or covalent?
In NiCl2 the Ni-Cl bonds have “ionic character“.
What is the hybridization of NiCl2?
The NICI2 has sp-3 hybridization structure having trigonal hybridization shape but due to the presence of lone pair of electron an iodine atom the structure is distorted.
Which primary colors does nickel II chloride absorb?
Sodium Chloride was colorless so it absorbed all three of the primary colors. Cobalt (II) chloride appeared red so it absorbed green and blue. Nickel (II) chloride appeared green, so it absorbed blue and red.
Is NO2 ionic or molecular?
NO2 is covalent bond. because the electronegativity difference between the nitrogen and oxygen in NO2 is not enough to cause an ionic interaction,so it can’t be ionic bond.
How many ions does nickel have?
The outermost, or valence, electrons occupy cloud-like regions, or orbitals, and the two valence electrons of nickel atoms can be lost relatively easily to form two common ions. The loss of a single electron results in an ion, defined as an atom that has lost or gained electrons, with a +1 charge.
Is NH3 molecular or ionic?
NH3 is a covalent bond. This is because, Nitrogen and Hydrogen have shared the electron. The main difference between an ionic and covalent bond is ionic bond will donate or accept electron. Meanwhile, the covalent bond will share the electron.
How many chlorine atoms are in the molecule NiCl2?
As you can see, each molecule of NiCl2 has two chlorine ions, so each mole of NiCl2 will have 2 moles of chlorine ions.
What is the percent composition of NiCl2?
ElementSymbolMass PercentNiccolumNi24.694%ChlorineCl29.832%HydrogeniumH5.089%OxygeniumO40.388%
Is AlCl3 polar or nonpolar?
AlCl3 monomer is trigonal planar (similar to BF3), and it is non-polar. The dipole moments of each of the Al-Cl bond is directed at 120 degree angles to each other in a plane, and is therefore cancelled out. Therefore it is a non-polar molecule.
Is H2O polar or nonpolar?
Water (H2O), like hydrogen fluoride (HF), is a polar covalent molecule. When you look at a diagram of water (see Fig. 3-2), you can see that the two hydrogen atoms are not evenly distributed around the oxygen atom.
Is NiS soluble in water?
Compound FormulaNiSMelting Point797 °CBoiling Point1388 °CDensity5.8 g/cm3Solubility in H2OInsoluble
What is the Iupac name of nicl2 pph3 2?
Dichlorobis (triphenylphosphine) nickel (II)
What is the color of solid Ni H2NCH2CH2NH2 3 Cl2?
It is a green solid. What is H2NCH2CH2NH2? What is [Ni(en)3]Cl2 and describe its characteristics. Why did you have to put acetone into the [Ni(en)3]Cl2 solution?
How many moles are in 6h20?
The molar mass of CoCl2 is 129.83 g/mol The molar mass of H2O is 18.02 g/mol and the molar mass of 6H2O is (6)( 18.02 g/mol) = 108.12 g/mol The molar mass of CoCl2∙6H2O is 237.95 g/mol (The 6 in front of the H2O needs to be taken into account!)
How do you write NiCl3?
- Nickel(III) Chloride.
- Nickelic Chloride.
- NiCl3.
- 165.0524.