What is the name for SrBr2
Strontium bromide (SrBr2)
What is SrBr2 composed of?
NamesStructureCrystal structuretetragonalHazardsMain hazardsCorrosive
What is the name of LiF?
Lithium fluoride (LiF) is used chiefly as a fluxing agent in enamels and glasses.
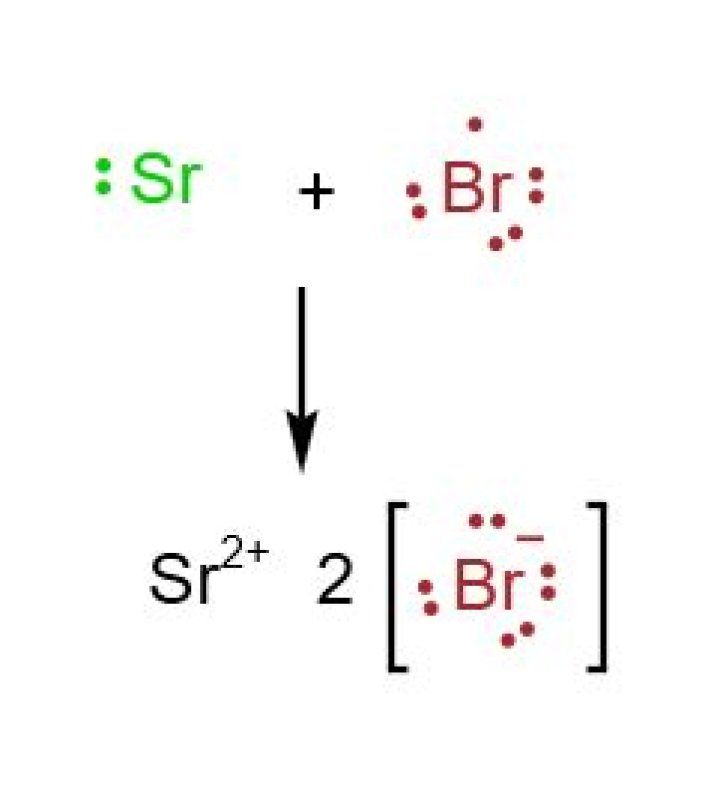
Why is SrBr2 an ionic compound?
So the compound is SRBR2. Strontium is not a transition metal, it’s just a regular metal so we just say it’s strontium. … Bromine is going to be your non metal and it has the base name of B R O M and remember that ending changes for your non metals, so it becomes I D E. So it’s bromide and not bromine.What is Al2S3 name?
Aluminum sulfide | Al2S3 | 99.9 | -100 mesh.
What is the name of Li2S?
Lithium sulfide (Li2S)
Is SrBr2 soluble or insoluble?
The anhydrate, SrBr2, is a colorless, transparent, crystal, odorless, having a bitter, saline taste. It is very deliquescent and soluble in water and is readily soluble in alcohol and amyl alcohol. The specific gravity of SrBr2 is 4.210 g/cm3.
Is NH4 a 2S?
Ammonium sulfide ((NH4)2S) exhibits high reactivity as a sulfide reagent in anion exchange reactions that transform CoO to cobalt sulfide nanoparticles (NPs).Is LiF ionic or covalent?
LiF is lithium fluoride. This is an example of a binary ionic compound, which consists of two elements, a cation and anion. Since lithium, the metal has a plus one charge, and fluoride, a nonmetal, has a negative charge, these two ions are held together through an ionic bond.
What is the correct Iupac name for CaBr2?PubChem CID24608StructureFind Similar StructuresMolecular FormulaBr2CaSynonymsCalcium bromide (CaBr2) calcium;dibromide Calcium bromide [JAN] Calcium bromide, ultra dry , 99.978% (metals basis) Calcium bromide (JAN) More…Molecular Weight199.89
Article first time published onWhat is the scientific name for NaBr?
Sodium bromide (NaBr)
What type of compound is BaSO4 What is its name?
Barium sulfate | BaSO4 – PubChem.
What is dilithium sulfide?
Lithium sulfide is the inorganic compound with the formula Li2S. It crystallizes in the antifluorite motif, described as the salt (Li+)2S2−. It forms a solid yellow-white deliquescent powder. In air, it easily hydrolyses to release hydrogen sulfide (rotten egg odor).
Why is calcium dichloride not the correct name for CaCl2?
CaCl is an ionic compound so the prefix system does not apply.
Is CaO a lime?
Calcium oxide (CaO), commonly known as quicklime or burnt lime, is a widely used chemical compound. It is a white, caustic, alkaline, crystalline solid at room temperature. … Calcium oxide that survives processing without reacting in building products such as cement is called free lime.
Why is Cl2 not dichloride?
Originally Answered: Why does chlorine exist as Cl2 but not CL? Because Cl2 is much more stable than just one chlorine atom. Cl has 7 electrons in its outer shell, meaning to obtain noble gas configuration (stable) it needs to gain one more electron.
Is LiF a molecule?
Li+ F−NamesIUPAC name Lithium fluorideIdentifiersCAS Number7789-24-4
Why is LiF polar?
The electronegativities of Li and F are more different (1.0 and 4.0) than those of H and F (2.1 and 4.0), so the LiF molecule is more polar than HF. … Li has a far greater tendency to lose its outer electron to F and to become a +1 ion than H does.
Is LiF an anion or cation?
MoleculeNameCationLiFlithium fluorideLiF+LiNe+NaLilithium sodiumNaLi+LiMgLithium magnesium
Is CaO a Chinese name?
Cao is the pinyin romanization of the Chinese surname 曹 (Cáo). It is listed 26th in the Song-era Hundred Family Surnames poem. Cao is romanized as Ts’ao in Wade-Giles, although the apostrophe is often omitted in practice.
What is the name for nh4no2?
Ammonium nitrite, NH4NO2, is the ammonium salt of nitrous acid.
What is NH4 2O called?
Chemical formula of ammonium oxide is (NH4)2O.
Is nh42s ionic or covalent?
The compound (NH4)2S ( N H 4 ) 2 S is formed from the cation ammonium (NH+4 N H 4 + ) and the sulfide anion (S2− ). Compounds made from cations and anions are ionic compounds.



