What is Z in Bohrs radius
Atomic number, Z is equal to 1. Hence the radius of nth orbit, rn = 0.529n2 Å.
What is Z in energy of electron?
-13.6 -Z eV/atom (Z = The energy of an electron moving in nth Bohr’s orbit of an element is given by E. =- atomic number).
What is E in Bohr model?
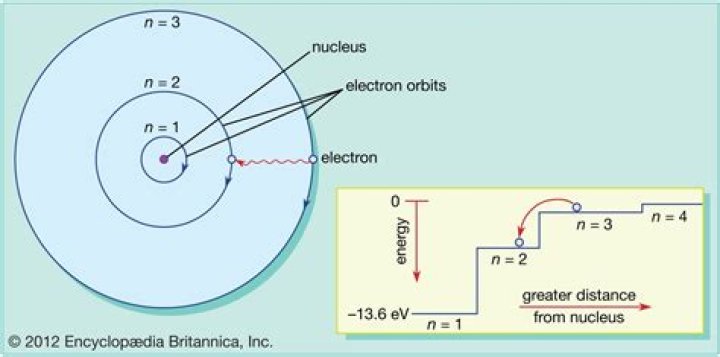
Bohr correctly proposed that the energy and radii of the orbits of electrons in atoms are quantized, with energy for transitions between orbits given by ∆E = hf = Ei − Ef, where ∆E is the change in energy between the initial and final orbits and hf is the energy of an absorbed or emitted photon.
What is Z in the Rydberg equation?
Rydberg formula Z is the atomic number (for hydrogen Z = 1 ), n₁ is the principal quantum number of the initial state (initial energy level), n₂ is the principal quantum number of the final state (final energy level), R is the Rydberg constant for hydrogen R ≈ 1.0973 * 10^7 1/m .What is the radius of first Bohr orbit for z 5?
The radius of first Bohr orbit of hydrogen atom is 0.529 A˚.
What is ze in chemistry?
Main characteristics of nuclei. … The charge of an atomic nucleus q is equal to minus the multiple of the absolute value of the electron charge -e, i.e., q = Ze. The integer Z determines the position of the atom in the periodic table, it is called the atomic number.
What is the ground state energy in eV of a hydrogen like atom with Z 2?
For Hydrogenic atoms: EI’ = Z2EI(μ’/μ) . If (μ’/μ) ~ 1, then EI’ = Z213.6 eV. For non-interacting electrons and Z = 2 we have EI’ = 54.4 eV.
What is kQ1Q2 R?
The force between charges Q1 and Q2 separated by a distance r is given by Coulomb’s Law: F = kQ1Q2 / r2, where k is a constant. F is attractive if the signs of charge are opposite and it is repulsive if the signs of charge are the same.What is K in kZe2 r2?
The attractive Coulomb force F = kZe2/r2, where k = (4peo)-1 = 9 x 109 Nm2/C2, balances mev2/r, which is the mass of the electron, me = 9.1×10-31kg, times the required acceleration to the center, v2/r.
What is Lyman Balmer and Paschen series?The Balmer series releases light in the visible region of the electromagnetic spectrum. The Lyman series, with longer arrows, requires the higher energy of the UV region. The Paschen and Brackett series, with shorter arrows require the lower energy of the IR region. Below is the visible emission spectrum of hydrogen.
Article first time published onWhat is n1 and n2 in Rydberg formula?
n1 and n2 are integers and n2 is always greater than n1. The modern value of Rydberg constant is known as 109677.57 cm-1 and it is the most accurate physical constant. According to Paschen series, n1 = 3 and n2 = 4, 5… λ = 1.282 x 10-4 cm = 1282 nm which is in near infrared region.
What was JJ Thomson's atomic model?
Summary. J.J. Thomson’s experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons. Thomson proposed the plum pudding model of the atom, which had negatively-charged electrons embedded within a positively-charged “soup.”
Who discovered the proton?
It is 100 years since Ernest Rutherford published his results proving the existence of the proton. For decades, the proton was considered an elementary particle.
What is the radius of first orbit of He+?
${{a}_{o}}$ is the radius of the first orbit of the hydrogen atom and is equal to 52.9 pm.
What is the radius of the first Bohr orbit in He+?
The radius of the first Bohr orbit of hydrogen atom is 0.52.
What is the radius of 5th orbit of hydrogen atom?
– Therefore the energy associated with the 5th orbit of the hydrogen atom is $-8.68\times {{10}^{-20}}$ J/atom . – Therefore the radius of the 5th Bhor’s orbit of the hydrogen atom is 1.3225 nm or 13.225$\overset{o}{\mathop{\text{A}}}\,$.
What is the energy of a hydrogen atom in ground state in joules?
Energy of an electron in the ground state of the hydrogen atom is −2. 18×10−18J.
How does the Bohr model of the hydrogen atom explain the hydrogen emission spectrum?
Bohr’s model explains the spectral lines of the hydrogen atomic emission spectrum. While the electron of the atom remains in the ground state, its energy is unchanged. When the atom absorbs one or more quanta of energy, the electron moves from the ground state orbit to an excited state orbit that is further away.
What is the energy in Joules and the wavelength in meters of the photon produced when an electron falls from the N 5 to the N 3 level in a He+ ion Z 2 for He +)?
What is the energy in joules and the wavelength in meters of the photon produced when an electron falls from the n = 5 to the n = 3 level in a He+ ion (Z = 2 for He+)? Answer: 6.198 × 10–19 J; 3.205 × 10−7 m.
What is Z and A in chemistry?
Z = atomic number = number of protons in the nucleus = number of electrons orbiting the nucleus; A = mass number = number of protons and neutrons in the most common (or most stable) nucleus.
Which element is Z?
Zirconium – Element information, properties and uses | Periodic Table.
What does Ze mean in physics?
Neutrons carries no charge and protons carries positive charge. Thus, the total charge of the nucleus is due to protons only. As the number of protons of the nucleus is equal to the atomic number of atom ie. If the charge on each proton is e. Then, the total charge of the nucleus will be Ze.
What is E in atomic structure?
The development of modern atomic theory revealed much about the inner structure of atoms. … When describing the properties of tiny objects such as atoms, we use appropriately small units of measure, such as the atomic mass unit (amu) and the fundamental unit of charge (e).
What is kq1q2 r2?
Description. Named after Charles-Augustin de Coulomb, Coulomb’s Law (F=kqq/r2) is an equation that represents the attractive or repulsive electric force (F) of two point charges (q). … This causes the motion of particles and is measured as the electric force.
What is q1 and q2 in potential energy?
Consider the situation in QQ3 again: q1 is a negative source. charge and q2 a test charge. When q2 is changed from. positive to negative, the potential energy of the two-charge.
What is the value of k in F kq1q2 R 2?
The force between charges. The force exerted by one charge q on another charge Q a distance r away is given by: Coulomb’s law: F = kqQ / r2. the constant k = 8.99 x 109 N m2 / C2.
Does Balmer and Paschen overlap?
(C) No 2 series (e.g. Lyman, Balmer, Paschen etc.) overlap with each other, i.e. lowest frequency of one series will be greater than highest frequency of the next series.
What do you mean by Paschen series in physics?
In physics, the Paschen series (also called Ritz-Paschen series) is the series of transitions and resulting emission lines of the hydrogen atom as an electron goes from n ≥ 4 to n = 3, where n refers to the principal quantum number of the electron.
Is Paschen series ultraviolet?
The Lyman series is in the ultraviolet while the Balmer series is in the visible and the Paschen, Brackett, Pfund, and Humphreys series are in the infrared.
How do you find n1 and n2?
The inverse ratio relation is also true if we consider N1 & N2, as rotational speeds (rpm) of drive gear & driven gear resp. and T1 & T2 are torques for drive gear & driven gear resp. The relation is: N2 / N1 = T1 / T2.
What is n1 and n2 in chemistry?
Re: Question about n1 and n2 Yes n1 is the initial energy level and n2 is the final energy level.